|
This creates an intense repulsive electric field. The problem in the fusion of heavy nuclei is the large numbers of protons in both nuclei. Imagine touching two droplets of water – they will “snap together” because of surface tension to form a combined larger droplet. How were these new elements created on Earth?Ītoms of superheavy elements are made by nuclear fusion. But longer-lived (more neutron-rich) superheavy nuclei are expected to be situated at the centre of the so-called “ island of stability”, a place where neutron-rich nuclei with extremely long half-lives should exist.Ĭurrently, the isotopes of new elements that have been discovered are on the “shore” of this island, since we cannot yet reach the centre. Those superheavy nuclei that have been created artificially have decay lifetimes between nanoseconds and minutes. They are not found in nature, because they undergo radioactive decay to lighter elements. 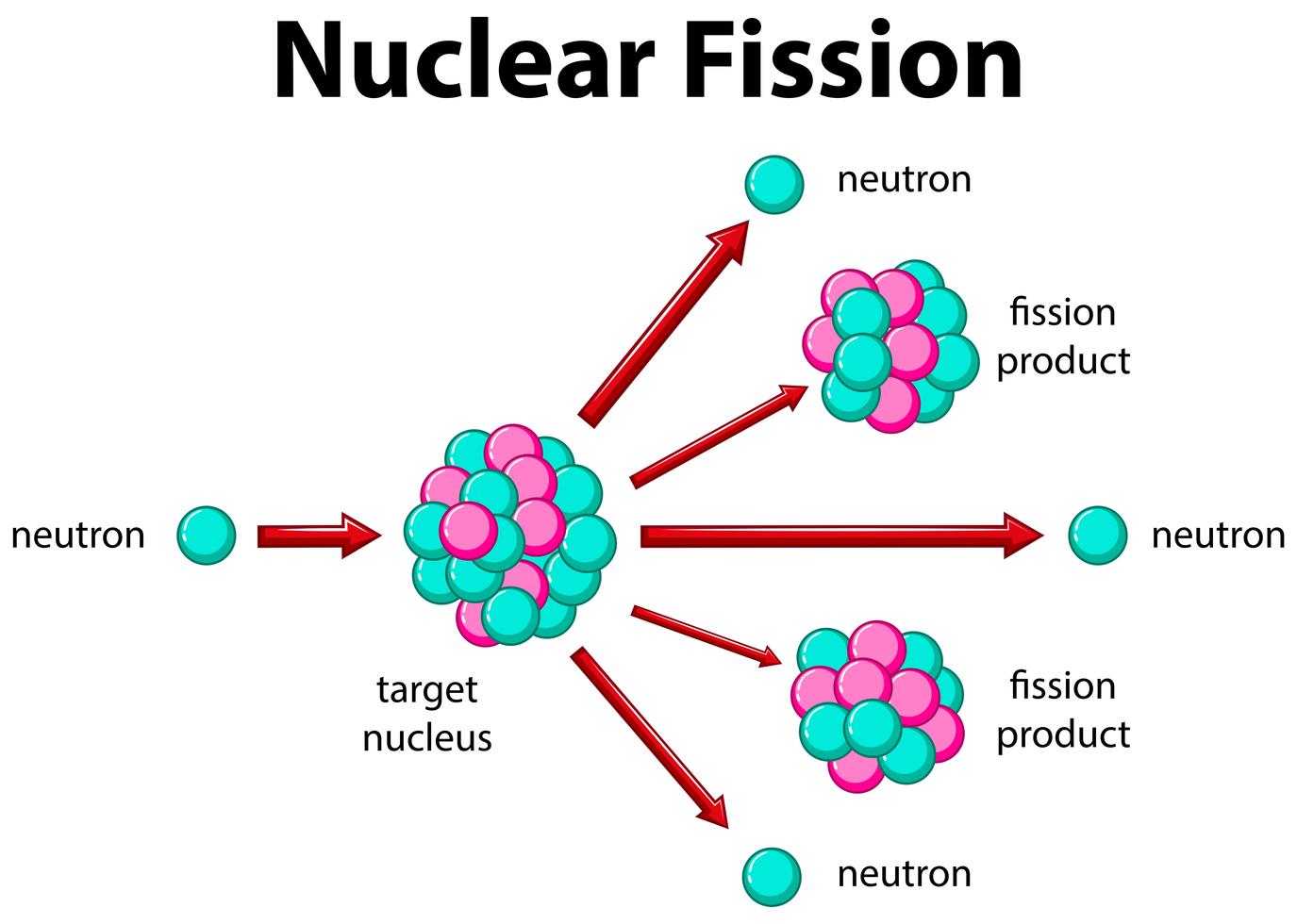
The superheavy elementsĮlements heavier than Rutherfordium (element 104) are referred to as superheavy. These names are not actually used by physicists, who instead refer to them as “element 118”, for example. 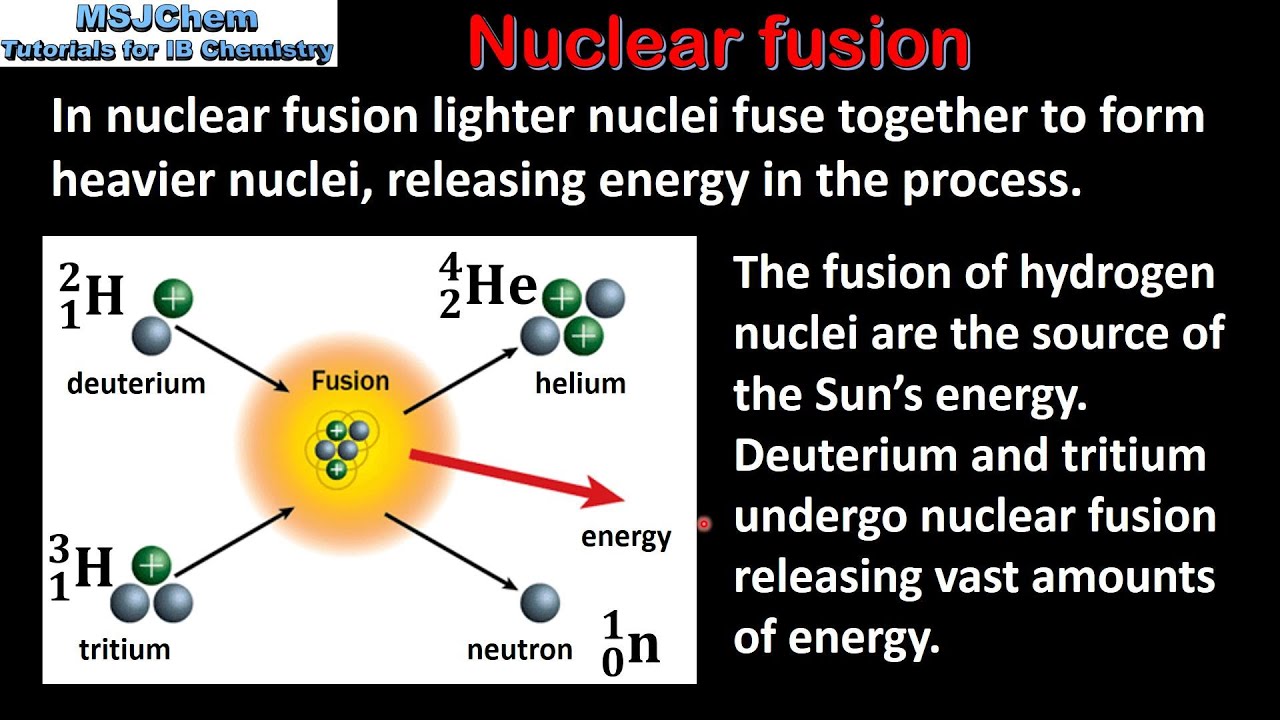
Element 113 is known as ununtrium (Uut), 115 is ununpentium (Uup), 117 is ununseptium (Uus) and 118 ununoctium (Uuo). Until their discovery and naming, all superheavy elements (up to 999!) have been assigned temporary names by the IUPAC. These names will be assessed by IUPAC, and once approved, will become the new names that scientists and students will have to remember. The joint Russian/US teams will make suggestions for elements 115, 117 and 118. The Japanese team will suggest the name for element 113. This completes the seventh row of the periodic table, and means that all elements between hydrogen (having only one proton in its nucleus) and element 118 (having 118 protons) are now officially discovered.Īfter the excitement of the discovery, the scientists now have the naming rights. The completed seventh row in the periodic table. Late last year, on December 30, 2015, IUPAC announced that claims for the discovery of all four new elements had been accepted. 
The measurements had been made at Nuclear Physics Accelerator laboratories in Russia (Dubna) and Japan (RIKEN) between 20. To add four in one go is quite an achievement but the race to find more is ongoing.īack in 2012, the International Unions of Pure and Applied Chemistry ( IUPAC) and Pure and Applied Physics ( IUPAP) tasked five independent scientists to assess claims made for the discovery of elements 113, 115, 117 and 118. In an event likely never to be repeated, four new superheavy elements were last week simultaneously added to the periodic table. Image showing periodicity of the chemical elements for enthalpy of fusion in a 3D periodic table column style.David Hinde, Australian National University Image showing periodicity of the chemical elements for enthalpy of fusion in a 3D spiral periodic table column style. Image showing periodicity of the chemical elements for enthalpy of fusion in a spiral periodic table heatscape style. Image showing periodicity of the chemical elements for enthalpy of fusion in a periodic table heatscape style. Image showing periodicity of the chemical elements for enthalpy of fusion in a periodic table cityscape style.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
